CAR-T Cell Therapy Shows Significant Efficacy in R/R CNSL Patients with TP53 Mutations
2026-2-13Relapsed/refractory central nervous system lymphoma (R/R CNSL) is associated with poor survival outcomes, which vary significantly depending on multiple factors. Among them, TP53 mutation is considered one of the key determinants of treatment response and has long posed a clinical challenge. In recent years, chimeric antigen receptor T-cell (CAR-T) therapy has demonstrated promising efficacy and safety in R/R CNSL. However, limited data are available regarding whether TP53 mutations influence the effectiveness of CAR-T therapy, warranting further investigation.
Recently, the lymphoma and myeloma team at GoBroad Healthcare Group, led by Professor Ke Xiaoyan and Professor Hu Kai, published a study entitled “CAR-T cell therapy in TP53-mutated CNS lymphoma: overcoming a high-risk genetic barrier” in Frontiers in Medicine. The first author of the paper is Li Danyang. This study provides new insights into CAR-T therapy for TP53-mutated R/R CNSL.
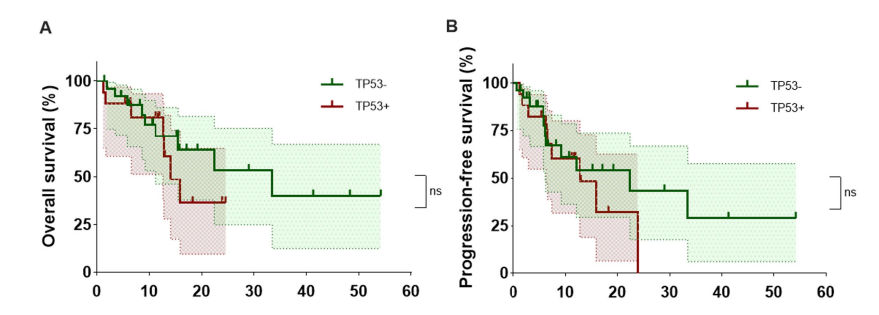
Professor Hu Kai explained that the treatment of R/R CNSL remains challenging. Patients who are sensitive to chemotherapy may achieve remission and benefit from autologous hematopoietic stem cell transplantation. However, in real-world practice, many patients relapse and develop chemotherapy resistance, particularly those harboring TP53 mutations, resulting in poor prognosis. The team retrospectively analyzed 61 patients with R/R CNSL, 43 of whom received CAR-T therapy. Overall survival (OS) and progression-free survival (PFS) were compared between patients with TP53 mutations (TP53+) and those with wild-type TP53 (TP53−), and prognostic factors were evaluated.
Key Findings
1. Comparable efficacy of CAR-T therapy in TP53+ and TP53− R/R CNSL patients In the TP53+ group, the overall response rate (ORR) and complete response rate (CRR) were both 64.5%. In the TP53− group, the ORR and CRR were 73.3% and 69.2%, respectively, indicating similar response rates between the two groups. The median PFS in the TP53+ group was 12.77 months (95% CI: 6.33–∞), compared with 22.4 months (95% CI: 6.13–∞) in the TP53− group. Although numerically shorter, the difference was not statistically significant. These results suggest that CAR-T therapy is effective in R/R CNSL patients with TP53 mutations.
2. Potentially greater benefit in non-GCB TP53+ R/R CNSL patients Subgroup analysis demonstrated that the cell-of-origin (COO) classification significantly influenced long-term survival. In the TP53+ cohort, patients with the non-germinal center B-cell-like (non-GCB) subtype had significantly longer OS than those with the GCB subtype (P = 0.003).
The preliminary findings from this center indicate that CAR-T therapy represents an effective treatment option for TP53-mutated R/R CNSL patients and may offer particular benefit for those with the non-GCB subtype. These data provide valuable clinical reference for treatment selection.
Conclusion and Future Perspectives
Professor Ke Xiaoyan noted that the lymphoma and myeloma department at GoBroad Healthcare Group has accumulated extensive experience in CAR-T therapy. As the number of real-world CAR-T cases increases, clinical management has become more complex. Clinical experience suggests that patients with TP53 mutations often exhibit high resistance to chemotherapy, whereas CAR-T therapy may provide meaningful benefit. Optimizing the timing of CAR-T therapy and implementing individualized, refined, and comprehensive management strategies remain key objectives in hematologic oncology.
Future approaches may integrate clinical, pathological, molecular, and imaging diagnostics to identify R/R CNSL patients who are resistant to conventional therapies but likely to benefit from CAR-T treatment. In addition, monitoring circulating tumor DNA (ctDNA) may enable earlier detection of relapse and timely intervention, ultimately prolonging survival.














